March 28, 2019
Probe shines light on overactive immune cells to help detect, treat certain cancers, autoimmune diseases
 A probe technology from Purdue University may help detect and treat certain cancers and autoimmune diseases. (Stock photo)
Download image
A probe technology from Purdue University may help detect and treat certain cancers and autoimmune diseases. (Stock photo)
Download image
WEST LAFAYETTE, Ind. – One frustration for doctors and patients dealing with certain cancers and autoimmune diseases is that they know the cause, but they don’t know how to reduce its effects in the body.
A clue can be found with the immunoproteasome, a protein complex in the body that is present in specific types of cancers, such as colorectal, colon and breast. It also is present in autoimmune diseases where cells have encountered a pro-inflammatory signal where the body’s immune cells start to attack and destroy themselves. Identifying and reducing this autoimmune response is an ongoing challenge for medical professionals.
“It is well understood which types of cells will express the immunoproteasome,” said Darci Trader, an assistant professor of medicinal chemistry and molecular pharmacology in Purdue’s College of Pharmacy, who leads the research team. “How its activity can be perturbed to affect disease is not.”
Trader and her Purdue University team have designed a new probe to monitor and label immunoproteasome-expressing cells.
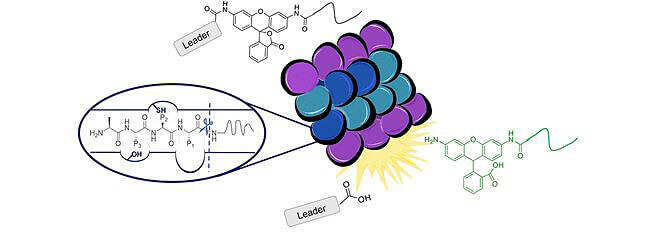 Purdue University researchers have developed a new probe to monitor and label immunoproteasome-expressing cells. (Image provided)
Download image
Purdue University researchers have developed a new probe to monitor and label immunoproteasome-expressing cells. (Image provided)
Download image
Current probes are inefficient for monitoring the activity of the immunoproteasome in live cells because they are not very cell permeable and can easily be degraded by other cellular proteases. The work is published in the March 20 edition of the Journal of the American Chemical Society.
“Having an immunoproteasome-selective probe that can work in live cells allows for a quicker readout of how small molecules are affecting the activity of the immunoproteasome,” Trader said. “We are going to use our new immunoproteasome probe to discover small molecules to help tweak the immune system response for people dealing with cancers, infections and autoimmune diseases.”
In the case of cancer or infections, the aim is a quicker immune system response. In the case of autoimmune diseases, the goal is to lessen the immune systems response. As the probe fluoresces to indicate immunoproteasome is present, the identification of small molecules could lead to increasing or decreasing the immune system response to best serve the patient.
Trader said they also are working to test the ability of the probe to detect the immunoproteasome and then use it as a vehicle to release drugs to treat cancer and other diseases. They are specifically looking at the use of prodrugs, which are biologically inactive compounds that selectively produce medication effects based on how they are created.
Their work aligns with Purdue's Giant Leaps celebration, celebrating the global advancements in health as part of Purdue’s 150th anniversary. Health is one of the four themes of the yearlong celebration’s Ideas Festival, designed to showcase Purdue as an intellectual center solving real-world issues.
Trader has worked with the Purdue Office of Technology Commercialization to patent several technologies to help in disease monitoring and treatment. Her research team is looking for additional partners and researchers.
About Purdue Office of Technology Commercialization
The Purdue Office of Technology Commercialization operates one of the most comprehensive technology transfer programs among leading research universities in the U.S. Services provided by this office support the economic development initiatives of Purdue University and benefit the university's academic activities. The office is managed by the Purdue Research Foundation, which received the 2016 Innovation and Economic Prosperity Universities Award for Innovation from the Association of Public and Land-grant Universities. For more information about funding and investment opportunities in startups based on a Purdue innovation, contact the Purdue Foundry at foundry@prf.org. For more information on licensing a Purdue innovation, contact the Office of Technology Commercialization at otcip@prf.org. The Purdue Research Foundation is a private, nonprofit foundation created to advance the mission of Purdue University.
Writer: Chris Adam, 765-588-3341, cladam@prf.org
Source: Darci Trader, dtrader@purdue.edu
Abstract
Monitoring the Immunoproteasome in Live Cells Using an Activity-Based Peptide–Peptoid Hybrid Probe
Breanna L. Zerfas and Darci J. Trader
Activity-based probes have greatly improved our understanding of the intrinsic roles and expression levels of various proteins within cells. To be useful in live cells, probes must be cell permeable and provide a read-out that can be measured without disrupting the cells or the activity of the target. Unfortunately, probes for the various forms of the proteasome that can be utilized in intact cells are limited; commercially available probes are most effectively used with purified protein or cell lysate. The proteasome, both the 26S and various isoforms of the 20S CP, is an important target with reported roles in cancer, autoimmune disorders, and neurodegenerative diseases. Here, we present the development of a selective probe for the immunoproteasome, a specialized isoform of the 20S proteasome, that becomes expressed in cells that encounter an inflammatory signal. Using a one-bead, one-compound library of small peptides, we discovered a trimer sequence efficiently cleaved by the immunoproteasome with significant selectivity over the standard proteasome. Upon conjugating this sequence to rhodamine 110 and a peptoid, we generated a probe with a considerable improvement in sensitivity compared to that of current aminomethylcoumarin-based proteasome probes. Importantly, our probe was capable of labeling immunoproteasome-expressing cells while maintaining its selectivity over other cellular proteases in live cell cultures. We anticipate this probe to find wide utility for those that wish to study the immunoproteasome’s activity in a variety of cell lines and to be used as a reporter to discover small molecules that can perturb the activity of this proteasome isoform.

