January 22, 2016
Two cells are better than one at sensing the environment, but five is a crowd
 |
|
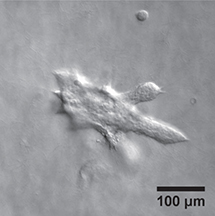
This image shows a mouse mammary epithelial cell grouping, or "organoid," with branching protrusions to the side with higher concentrations of epidermal growth factor. (Purdue University photo/courtesy of Andrew Mugler) |
WEST LAFAYETTE, Ind. — As cells group together they can more precisely measure changes in their environment, but the increase in ability plateaus after the group exceeds three to four cells, a team of scientists report.
The team developed a new theory of cellular collective gradient sensing from the study of mouse mammary cells that must sense and react to pregnancy-related hormones, said Andrew Mugler, an assistant professor of physics involved in the research.
"Single cells can sense their environment with remarkable precision, for instance amoebas respond to changes in chemical concentrations of just one percent across the cell body, but groups of cells are even better and can detect changes in chemical concentrations of fractions of a percent," said Mugler, who is part of Purdue University's Department of Physics and Astronomy. "We wanted to find out how the cells coordinate and collectively behave to achieve this improvement. We found that cellular communication is the key to the improved sensitivity, but it has its limits."
Mugler is currently working with an experimental group at Purdue to apply the theoretical framework to the collective behavior of metastatic breast cancer cells.
"The theoretical framework and model of collective sensing could be applied to any cellular system that uses molecule exchange to collectively sense its environment, including embryonic development or cancer metastasis," he said. "We are eager to tap into its potential to support research into prevention and treatment of disease."
Mugler was part of a team that included scientists from Emory University, Yale University and Johns Hopkins University.
Two papers detailing the experimental results and new theoretical framework for the collective behavior were published in the Proceedings of the National Academy of Sciences and are available online.
The team studied the response of the cells to variations in the concentration, or gradients, of epidermal growth factor. Groups of the cells, which scientists in the field call "organoids" because they are too few to form a complete tissue or organ, were placed in a collagen-filled container. The concentration of epidermal growth factor increased across the container and the gradient was kept below the limit of what could be detected by a single cell.
Detection of the difference in concentration of the growth factor was determined by the growth of branch-like protrusions from the organoid that are the precursor to cellular movement or growth. A significant increase in these protrusions on the side with a higher concentration of growth factor meant the cells could detect the difference, and purely random growth of the protrusions meant they could not. The steeper the difference in gradient from one side of the organoid to the other, the more biased the branching was to the higher side, Mugler said.
"It is quite clear the cells are working together and each is not simply detecting and acting upon the information from its immediate environment," he said. "The cells on the outside edge of the group have access to the most information about the gradient, but somehow the cells were exchanging their individual measurements among the group. Each cell would need a global and local readout of the environment in order to decide whether or not to move in that direction."
The team tested the role of cellular communication by adding a drug that blocks junctions between neighboring cells used to exchange molecules. They found that without this communication the cells no longer responded to the difference in gradient. They also were able to determine that the communication involved the release of calcium molecules, he said.
"The information is being relayed from one cell to its neighboring cell, like the game of telephone," Mugler said. "Like the game, the information being shared among the cells degrades after a certain point. We found that the sensitivity to the gradient was enhanced as additional cells were added, but plateaued at around three to four cells, suggesting this is as far as the messages travel."
Previous theories of cellular gradient sensing did not account for the game-of-telephone-like relay of information and predicted a continuous increase in sensitivity as the number of cells in the group increased, he said.
After determining calcium release was involved in the cellular communication, the team performed an experiment in which these molecules were illuminated. The communication could be seen as the cells lit up and dimmed in neighborhoods of only three or four cells, Mugler said.
Mugler and Ilya Nemenman, a theoretical biophysicist at Emory, led the theoretical side of the research team and developed the mathematical models. The work began when Mugler was a postdoctoral fellow in Nemenman's laboratory and it continued after he became part of Purdue's faculty.
The experiments were conducted primarily at Johns Hopkins and led by Andre Levchenko, a biomedical engineer at Yale who studies how cells navigate, and Andrew Ewald, a biologist at Johns Hopkins who studies the cellular mechanisms of cancer.
The National Science Foundation, the National Institutes of Health, James S. McDonnell Foundation and Human Frontiers Science Program funded the research.
Writer: Elizabeth K. Gardner, 765-494-2081, ekgardner@purdue.edu
Source: Andrew Mugler, 765-496-3427, amugler@purdue.edu
Link to Emory news release: http://esciencecommons.blogspot.com
Abstract
Cell-cell communication enhances the capacity of cell ensembles to sense shallow gradients during morphogenesis
David Ellison, Andrew Mugler, Matthew Brennan, Sung Hoon Lee, Robert Huebner, Eliah Shamir, Laura A. Woo, Joseph Kim, Patrick Amar, Ilya Nemenman, Andrew J. Ewald, Andre Levchenko
Collective cell responses to exogenous cues depend on cell-cell interactions. In principle, these can result in enhanced sensitivity to weak and noisy stimuli. However, this has not yet been shown experimentally, and little is known about how multicellular signal processing modulates single cell sensitivity to extracellular signaling inputs, including those guiding complex changes in the tissue form and function. Here we explored if cell-cell communication can enhance the ability of cell ensembles to sense and respond to weak gradients of chemotactic cues. Using a combination of experiments with mammary epithelial cells and mathematical modeling, we find that multicellular sensing enables detection of and response to shallow Epidermal Growth Factor (EGF) gradients that are undetectable by single cells. However, the advantage of this type of gradient sensing is limited by the noisiness of the signaling relay, necessary to integrate spatially distributed ligand concentration information. We calculate the fundamental sensory limits imposed by this communication noise and combine them with the experimental data to estimate the effective size of multicellular sensory groups involved in gradient sensing. Functional experiments strongly implicated intercellular communication through gap junctions and calcium release from intracellular stores as mediators of collective gradient sensing. The resulting integrative analysis provides a framework for understanding the advantages and limitations of sensory information processing by relays of chemically coupled cells.
Abstract
Limits to the precision of gradient sensing with spatial communication and temporal
integration
Andrew Mugler, Andre Levchenko, and Ilya Nemenman
Gradient sensing requires at least two measurements at different points in space. These measurements must then be communicated to a common location to be compared, which is unavoidably noisy. While much is known about the limits of measurement precision by cells, the limits placed by the communication are not understood. Motivated by recent experiments, we derive the fundamental limits to the precision of gradient sensing in a multicellular system, accounting for communication and temporal integration. The gradient is estimated by comparing a "local" and a "global" molecular reporter of the external concentration, where the global reporter is exchanged between neighboring cells. Using the fluctuation-dissipation framework, we find, in contrast to the case when communication is ignored, that precision saturates with the number of cells independently of the measurement time duration, since communication establishes a maximum lengthscale over which sensory information can be reliably conveyed. Surprisingly, we also find that precision is improved if the local reporter is exchanged between cells as well, albeit more slowly than the global reporter. The reason is that while exchange of the local reporter weakens the comparison, it decreases the measurement noise. We term such a model "regional excitation–global inhibition" (REGI). Our results demonstrate that fundamental sensing limits are necessarily sharpened when the need to communicate information is taken into account.

