August 10, 2016
Research suggests new tool for cancer treatment based on cell type
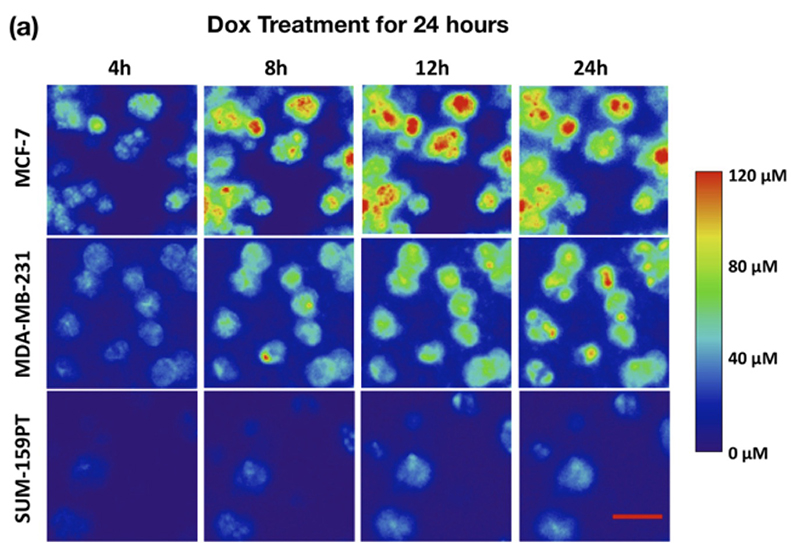 This image shows how three different types of human breast cancer cells react to a chemotherapy drug, taking up the drug at differing rates, by using a new tumor-modelling platform called a tumor-microenvironment-on-chip (T-MOC). Red means more drug uptake. (Purdue University photo/Bumsoo Han)
Download image
This image shows how three different types of human breast cancer cells react to a chemotherapy drug, taking up the drug at differing rates, by using a new tumor-modelling platform called a tumor-microenvironment-on-chip (T-MOC). Red means more drug uptake. (Purdue University photo/Bumsoo Han)
Download image
WEST LAFAYETTE, Ind. – A new tumor model has been shown to predict how certain types of cancer cells react differently to a commonly used chemotherapy drug, a potential tool for "precision medicine," in which drug treatment is tailored to individual patients and certain cancer types.
Drug resistance and various subtypes of tumors represent critical bottlenecks for effective chemotherapy.
"This means rapid and accurate screening of effective drugs and drug combinations can be extremely useful to realize precision medicine for cancer therapy," said Bumsoo Han, a Purdue University professor of mechanical and biomedical engineering
Recent research findings demonstrate that a new tumor-modeling platform called a tumor-microenvironment-on-chip (T-MOC) can be used to visualize and quantify how different types of cancer cells react differently to the chemotherapy drug doxorubicin. The drug sometimes failed during testing to kill cancer cells yet stopped their growth.
"So, there are differences in the response to the drug based on cancer type," Han said. "Our platform makes it possible to assess the efficacy of chemotherapeutic drugs according to cell-type; for example, which cells actually died from this drug, which cells didn't die but stopped actively growing, or even which cells survived the drug."
Based on experimental data, the researchers developed a theoretical model to quantify and ultimately predict the effectiveness of drugs depending on the type of cancer cell being treated.
The findings were detailed in a paper published in the July issue of Molecular Pharmaceutics. The paper was authored by graduate student Kyeonggon Shin, undergraduate Brett S. Klosterhoff and Han.
Researchers performed experiments using the T-MOC platform that could help usher in precision medicine, in which drug treatment is tailored to specific patients and certain cell types.
"We are providing a new tool, a new platform to test drugs and drug delivery systems for precision medicine," Han said.
The device is about 4.5 centimeters (1.8 inches) square and contains "microfluidic" channels where cancer cells were cultured within a three-dimensional "extracellular matrix," a scaffold-like material found between cells in living tissue. The experiments also incorporated "interstitial fluid," which is found inside tumors and is thought to be a barrier to drug delivery.
Three types of human breast cancer cell lines were cultured on the T-MOC platform, and their drug response and resistance to doxorubicin were characterized by time-lapse fluorescence microscopy.
"Particularly in breast cancer, there are different subgroups of cancers," Han said. "Depending on the subgroup, the drug response is different. So, to study the efficacy of a drug and its nanoparticle formulation, we examined how the drug and nanoparticles were transported through the interstitial space and uptaken by different types of cancer cells."
Two of the three types of breast cancer cells tested were triple negative breast cancer, one of the most-difficult-to-treat forms of the disease.
"Even within triple negative there are additional subtypes, so this is not one single disease," Han said. "The targeting drugs only work with certain types of cells."
The findings also showed some cell types take up drugs more rapidly than others.
"You can see that, depending on the cell type, the response to the same drug is different and the drug uptake is different," he said.
Future research will involve comparing and validating research results with data from laboratory animal tests and incorporating a more complex mixture of tumor stromal components in the T-MOC experiments. Ultimately, in precision medicine, a patient's tumor cells might be studied using a T-MOC system to determine the proper drug and dosage to use before beginning treatment, Han said.
Research to fabricate the microfluidic device was performed at the Birck Nanotechnology Center in Purdue's Discovery Park. The work also is associated with the Purdue University Center for Cancer Research.
The research was partially funded by the National Institutes of Health; a Collaboration in Translational Research Award from the Indiana Clinical and Translational Sciences Institute; the Walther Cancer Foundation; and the Digital Human Project.
Writer: Emil Venere, 765-494-4709, venere@purdue.edu
Source: Bumsoo Han, 765-494-5626, bumsoo@purdue.edu
ABSTRACT
Characterization of Cell-Type-Specific Drug Transport and Resistance of Breast Cancers Using Tumor-Microenvironment-on-Chip
Kyeonggon Shin,† Brett S. Klosterhoff,† and Bumsoo Han*
†School of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United States §Weldon School of Biomedical Engineering, Birck Nanotechnology Center, and Purdue Center for Cancer Research, Purdue University, West Lafayette, Indiana 47907, United States * Corresponding author, e-mail bumsoo@purdue.edu
Heterogeneous response and resistance of cancer cells to chemotherapeutic drugs pose a significant challenge for successful cancer treatments. In this study, an integrated experimental and theoretical analysis of cellular drug transport was developed. The experimental platform, called tumor-microenvironment-on-chip (T-MOC), is a microfluidic platform where cancer cells were cultured within a three-dimensional extracellular matrix perfused with interstitial fluid. Three types of human breast cancer cell lines (MCF-7, MDA-MB-231, and SUM-159PT) were cultured on this T-MOC platform, and their drug response and resistance to doxorubicin were characterized by time-lapse quantitative fluorescence microscopy. To study the effects of nanoparticle-mediated drug delivery, the transport and action of doxorubicin encapsulated nanoparticles were also examined. Based on the experimental data obtained, a theoretical model was developed to quantify and ultimately predict the cellular transport processes of drugs cell-type specifically. The results demonstrate that the cellular drug transport can be celltype-specifically quantified by rate constants representing the uptake and efflux of doxorubicin across the cellular membrane.

