November 11, 2019
Getting cancer drugs to the brain is difficult – but a new ‘road map’ might make it easier
Tiffany Lyle, assistant professor of veterinary anatomic pathology at Purdue University, led research that has provided the first comprehensive characterization of both the blood-brain and blood-tumor barriers in brain metastases of lung cancer, which will serve as a road map for treatment development. (Purdue University photo/Rebecca Wilcox)
WEST LAFAYETTE, Ind. — The human brain has some remarkable capabilities – including the ability to block cancer drugs from effectively reaching cancer cells in the brain.
The greatest obstacle when it comes to treating cancer that has spread to the brain is the blood-brain barrier, the brain’s natural defense mechanism that is a collection of blood vessels that can filter out what goes in and out of the brain.
Purdue University scientists have provided the first comprehensive characterization of both the blood-brain and blood-tumor barriers in brain metastases of lung cancer, which will serve as a road map for treatment development. The work was recently published in Oncotarget.
The research was led by Tiffany Lyle, assistant professor of veterinary anatomic pathology, whose work focuses on the pathology of the blood-brain barrier. As the principal investigator of the Comparative Blood-Brain Barrier Laboratory, she and her team have collaborated with scientists at Purdue and the Indiana University Simon Cancer Center.
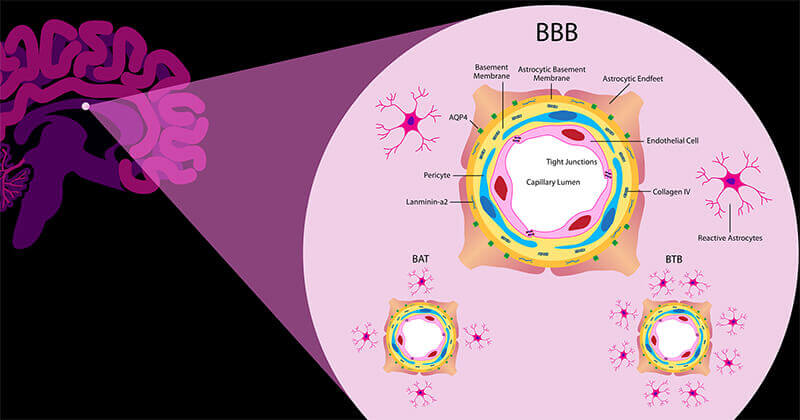 This illustration shows the components of the blood-brain barrier, a collection of blood vessels that filters what goes in and out of the brain. The blood-brain barrier is the greatest obstacle when it comes to treating cancer that has spread to the brain. (Image provided by Daniel Hertzberg)
Download image
This illustration shows the components of the blood-brain barrier, a collection of blood vessels that filters what goes in and out of the brain. The blood-brain barrier is the greatest obstacle when it comes to treating cancer that has spread to the brain. (Image provided by Daniel Hertzberg)
Download image
“Brain metastases occur most frequently in patients diagnosed with breast and lung cancer and melanoma,” Lyle said. “These metastases have a devastating survival rate, mostly because it’s so difficult to get drugs into the brain tissue because of the blood-brain barrier.”
Brain metastases, or secondary brain tumors, occur when cancer cells spread from their original site to the brain. This happens in 10% to 30% of adults with cancer, according to the Mayo Clinic.
When cancer cells invade the brain, the blood-brain barrier transitions into the blood-tumor barrier, and this transition still presents a roadblock for effective drug delivery to the brain. The formation of the blood-tumor barrier has been insufficiently characterized in lung cancer until now, Lyle said.
“We wanted to see what changes in the blood-brain barrier were occurring rapidly and which ones were sustained over time,” Lyle said. “Identifying those changes and pinpointing when they occur during the transition will be critical to developing treatment plans and being able to identify where, and when, cancer cells need to be targeted.”
The blood-brain and blood-tumor barriers were analyzed in animal models with non-small-cell cancer cells and immunofluorescent imaging. Researchers also validated their findings by studying the blood-tumor barrier of brain metastases in human post-mortem tissue.
Scientists observed that one of the sustained changes during the transition from the blood-brain barrier to the blood-tumor barrier was in one of the largest cell types in the brain that has numerous functions. These cells are known as astrocytes. Lyle said that discovery alone will be key when it comes to future treatment development.
“Identifying when that change occurs during the transition is critical because it tells us when and where the brain vasculature prevents effective drug delivery,” Lyle said.
Lyle also heads the Comparative Blood-Brain Barrier Laboratory at Purdue and is already collaborating with Yoon Yeo, a professor in the Department of Industrial and Physical Pharmacy, to work on improving drug delivery. Being able to truly target cancer drugs to diseased tissue could improve quality of life in patients, Lyle said.
“A goal of our research is to meaningfully contribute to the evolving field of personalized medicine and provide patients who have received a devastating diagnosis a sense of hope for treatment possibilities,” Lyle said.
The work was funded by the Showalter Foundation and the Indiana Clinical Translational Sciences Institute.
Writer: Abbey Nickel, 765-496-1325, nickela@purdue.edu
Source: Tiffany Lyle, lreed@purdue.edu
ABSTRACT
Dynamic transition of the blood-brain barrier in the development of non-small cell lung cancer brain metastases
Gozde Uzunalli, Alexandra M. Dieterly, Chinyere M. Kemet, Hsin-Yi Weng, Arvin H. Soepriatna, Craig J. Goergen, Aparna B. Shinde, Michael K. Wendt and L. Tiffany Lyle
Invasion of the brain by non-small cell lung cancer (NSCLC) results in a shift of the blood-brain barrier (BBB) to the insufficiently characterized blood-tumor barrier (BTB). Effective drug delivery through the BTB is one of the greatest therapeutic obstacles in treating brain metastases. Using an experimental model, we defined the key changes of the BTB and the BBB in the brain around the tumor (BAT) region over time. Brain-seeking NSCLC cells were delivered into the circulation of athymic-nude mice via intracardiac injection and developing brain metastases were evaluated over six-weeks. Components of the BBB and BTB were analyzed using immunofluorescence microscopy and compared using a mixed model of regression. Our results demonstrate a dynamic time-dependent BTB phenotype. Capillaries of the BAT and BTB were dilated with increased CD31 expression compared to controls. Expression of collagen IV, a pan-basement membrane component, was significantly decreased in the BTB compared to the BBB. There was also a significant increase in the desmin+ pericyte subpopulation in the BTB compared to the BBB. The most striking changes were identified in astrocyte water channels with a 12.18-fold (p < 0.001) decrease in aquaporin-4 in the BTB, the BAT was unchanged. Analysis of NSCLC brain metastases from patient samples similarly demonstrated dilated capillaries and loss of both collagen IV and aquaporin-4. These data provide a comprehensive analysis of the BTB in NSCLC brain metastasis. Astrocytic endfeet, pericytes, and the basement membrane are potential therapeutic targets to improve efficacy of chemotherapeutic delivery into NSCLC brain metastases.

