November 15, 2018
Targeted delivery: Purdue cancer identity technology makes it easier to find a tumor’s ‘address’
Improved drug delivery method is aimed at making chemotherapy easier to help treat people with various tumors.
WEST LAFAYETTE, Ind. – Purdue University researchers have developed a technology aimed at making it easier to deliver cancer treatment to the right “address” in the body while also easing the painful side effects of chemotherapy on patients.
One of the big issues with chemotherapy is that most treatment approaches focus on the tumor itself without paying significant attention to the microenvironment surrounding the tumor. The new method is detailed in the nanotechnology journal Small.
 An improved drug delivery method from Purdue University uses nanoparticles to help treat people with various tumors and ease the painful side effects of chemotherapy. (AP Photo)
Download image
An improved drug delivery method from Purdue University uses nanoparticles to help treat people with various tumors and ease the painful side effects of chemotherapy. (AP Photo)
Download image
“The traditional approach is similar to a delivery driver trying to drop off a package to a certain person without knowing their specific address,” said Yoon Yeo, a professor of industrial and physical pharmacy at Purdue, who is leading the research team. “Our new approach provides directions to find the specific address to deliver the chemotherapeutic drugs.”
The Centers for Disease Control and Prevention reports that each year, about 650,000 cancer patients receive chemotherapy in an outpatient oncology clinic in the United States. Patients receiving chemotherapy are at risk for various side effects that may lead to hospitalization, disruptions in chemotherapy schedules, and even death.
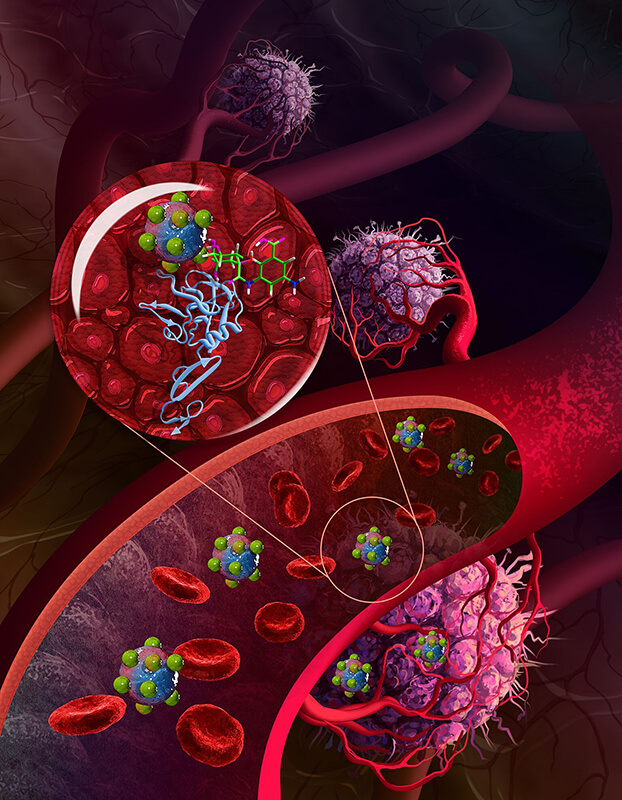 Purdue University researchers developed a technique to prepare polyol-modified nanoparticles so they locate cancerous cells and tumors by checking out blood vessels surrounding the tumors. (Image provided)
Download image
Purdue University researchers developed a technique to prepare polyol-modified nanoparticles so they locate cancerous cells and tumors by checking out blood vessels surrounding the tumors. (Image provided)
Download image
The Purdue method uses nanoparticles, which are considered promising carriers of drugs needed for chemotherapy to target tumors. The researchers developed a technique to prepare polyol-modified nanoparticles so they locate cancerous cells and tumors by checking out blood vessels surrounding the tumors.
The nanoparticles then interact with the vascular lining to enter tumors and destroy them. The Purdue researchers said their method helps the nanoparticles to exit from the circulation and enter tumors and better treat the cancer. They have tested the method on breast cancer and melanoma models and believe it also will prove effective for many types of cancerous tumors.
“Chemotherapy can be almost unbearable for most patients and we want to change that,” Yeo said. “Our method better targets tumors so lower dosages are required and the drugs do less damage to normal tissues.”
Their work aligns with Purdue's Giant Leaps celebration, acknowledging the university’s global advancements in health as part of Purdue’s 150th anniversary. This is one of the four themes of the yearlong celebration’s Ideas Festival, designed to showcase Purdue as an intellectual center solving real-world issues.
The technology is patented through the Purdue Office of Technology Commercialization, and the research team is looking for partners.
About Purdue Office of Technology Commercialization
The Purdue Office of Technology Commercialization operates one of the most comprehensive technology transfer programs among leading research universities in the U.S. Services provided by this office support the economic development initiatives of Purdue University and benefit the university's academic activities. The office is managed by the Purdue Research Foundation, which received the 2016 Innovation and Economic Prosperity Universities Award for Innovation from the Association of Public and Land-grant Universities. For more information about funding and investment opportunities in startups based on a Purdue innovation, contact the Purdue Foundry at foundry@prf.org. For more information on licensing a Purdue innovation, contact the Office of Technology Commercialization at otcip@prf.org. The Purdue Research Foundation is a private, nonprofit foundation created to advance the mission of Purdue University.
Writer: Chris Adam, 765-588-3341, cladam@prf.org
Source: Yoon Yeo, yyeo@purdue.edu
Abstract
Quinic Acid‐Conjugated Nanoparticles Enhance Drug Delivery to Solid Tumors via Interactions with Endothelial Selectins
Jun Xu, Steve Seung‐Young Lee, Howon Seo, Liang Pang, Yearin Jun, Ruo‐Yu Zhang, Zhong‐Yin Zhang, Pilhan Kim, Wooin Lee, Stephen J. Kron, Yoon Yeo
Current nanoparticle (NP) drug carriers mostly depend on the enhanced permeability and retention (EPR) effect for selective drug delivery to solid tumors. However, in the absence of a persistent EPR effect, the peritumoral endothelium can function as an access barrier to tumors and negatively affect the effectiveness of NPs. In recognition of the peritumoral endothelium as a potential barrier in drug delivery to tumors, poly (lactic‐co‐glycolic acid) (PLGA) NPs are modified with a quinic acid (QA) derivative, synthetic mimic of selectin ligands. QA‐decorated NPs (QA‐NP) interact with human umbilical vein endothelial cells expressing E‐/P‐selectins and induce transient increase in endothelial permeability to translocate across the layer. QA‐NP reach selectin‐upregulated tumors, achieving greater tumor accumulation and paclitaxel (PTX) delivery than polyethylene glycol‐decorated NPs (PEG‐NP). PTX‐loaded QA‐NP show greater anticancer efficacy than Taxol or PTX‐loaded PEG‐NP at the equivalent PTX dose in different animal models and dosing regimens. Repeated dosing of PTX‐loaded QA‐NP for two weeks results in complete tumor remission in 40–60% of MDA‐MB‐231 tumor‐bearing mice, while those receiving control treatments succumb to death. QA‐NP can exploit the interaction with selectin‐expressing peritumoral endothelium and deliver anticancer drugs to tumors to a greater extent than the level currently possible with the EPR effect.

