Research yields first detailed view of morphing Parkinson's protein
September 5, 2013
 |
|
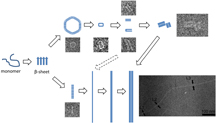
This graphic illustrates the morphing structure of a brain protein thought to play a role in Parkinson's disease, information that could aid the development of medications to treat the condition. Researchers have taken detailed images and measurements of the changing structure for the first time. Findings reveal that the protein morphs from its globular shape into "protofibril" strands that assemble into pore-like rings. These rings then open up, forming pairs of protofibrils that assemble into fibrils through hydrogen bonds. (Purdue University/ Hangyu Zhang) |
WEST LAFAYETTE, Ind. - Researchers have taken detailed images and measurements of the morphing structure of a brain protein thought to play a role in Parkinson's disease, information that could aid the development of medications to treat the condition.
The protein, called alpha synuclein (pronounced sine-yoo-cline), ordinarily exists in a globular shape. However, the protein morphs into harmful structures known as amyloid fibrils, which are linked to protein molecules that form in the brains of patients with neurodegenerative diseases.
"The abnormal protein formation characterizes a considerable number of human diseases, such as Alzheimer's, Parkinson's and Huntington's diseases and type II diabetes," said Lia Stanciu, an associate professor of materials engineering at Purdue University.
Until now, the transition from globular to fibrils had not been captured and measured.
Researchers incubated the protein in a laboratory and then used an electron microscope and a technique called cryoelectron microscopy to snap thousands of pictures over 24 hours, capturing its changing shape. The protein was frozen at specific time intervals with liquid nitrogen.
Findings reveal that the protein morphs from its globular shape into "protofibril" strands that assemble into pore-like rings. These rings then open up, forming pairs of protofibrils that assemble into fibrils through hydrogen bonds.
"We found a correlation between protofibrils in these rings and the fibrils, for the first time to our knowledge, by measuring their true sizes and visualizing the aggregation steps," Stanciu said. "A better understanding of the mechanism yields fresh insight into the pathogenesis of amyloid-related diseases and may provide us the opportunity to develop additional therapeutic strategies."
Parkinson's disease affects 1 percent to 2 percent of people older than 60, and an increase in its prevalence is anticipated in coming decades.
The findings were detailed in a research paper appearing in the June issue of the Biophysical Journal. The paper was authored by doctoral student Hangyu Zhang; former postdoctoral research associate Amy Griggs; Jean-Christophe Rochet, an associate professor of medicinal chemistry and molecular pharmacology; and Stanciu.
The researchers caused the protein to morph into fibrils by exposing it to copper, mimicking what happens when people are exposed to lead and other heavy metals. The contaminants interfere with the protein, changing the oxidation states of ions in its structure.
The research was funded by the National Institutes of Health. Future work will include experiments focusing on what happens when higher concentrations of copper are used.
Writer: Emil Venere, 765-494-4709, venere@purdue.edu
Source: Lia Stanciu, 765-496-3552, lastanciu@gmail.com
Note to Journalists: Journalists may obtain a copy of the research paper by contacting Emil Venere at 765-494-4709, venere@purdue.edu
ABSTRACT
In Vitro Study of a-Synuclein Protofibrils by Cryo-EM Suggests a Cu2D-Dependent Aggregation Pathway
Hangyu Zhang,† Amy Griggs,‡ Jean-Christophe Rochet,‡ and Lia A. Stanciu†§*
† Weldon School of Biomedical Engineering, ‡Department of Medicinal Chemistry and Molecular Pharmacology, and §School of Materials Engineering, Purdue University
The aggregation of a-synuclein is thought to play a role in the death of dopamine neurons in Parkinson's disease. Alpha-synuclein transitions itself through an aggregation pathway consisting of pathogenic species referred to as protofibrils (or oligomer), which ultimately convert to mature fibrils. The structural heterogeneity and instability of protofibrils has significantly impeded advances related to the understanding of their structural characteristics and the amyloid aggregation mystery. Here, we report, to our knowledge for the first time, on a-synuclein protofibril structural characteristics with cryoelectron microscopy. Statistical analysis of annular protofibrils revealed a constant wall thickness as a common feature. The visualization of the assembly steps enabled us to propose novel, to our knowledge, mechanisms for a-synuclein aggregation involving ring-opening and protofibril-protofibril interaction events. The ion channel-like protofibrils and their membrane permeability have also been found in other amyloid diseases, suggesting a common molecular mechanism of pathological aggregation. Our direct visualization of the aggregation pathway of a-synuclein opens up fresh opportunities to advance the understanding of protein aggregation mechanisms relevant to many amyloid diseases. In turn, this information would enable the development of additional therapeutic strategies aimed at suppressing toxic protofibrils of amyloid proteins involved in neurological disorders.

