Enzyme Catalysis
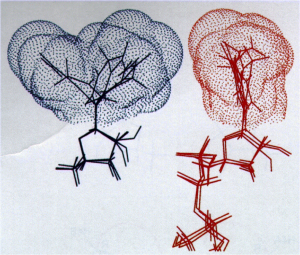 |
The Post group has worked to understand the basis of the catalytic power of enzymes using information from crystallography, kinetics, and a description of conformational distributions from molecular dynamics simulations. Instead of the enzyme mechanism described in biochemistry text-books, hen lysozyme hydrolysis was postulated to occur by a ring-opening mechanism based on substrate interactions defined by the active site geometry. In addition, an induced fit basis for substrate specificity of enzymes was rationalized from simple physical principles of enzyme kinetics. Of particular note is the proposal that ‘entropic guidance’ contributes to catalysis of NADH hydride transfer by dehydrogenases. Entropic guidance was suggested based on spatial constraints of thermal fluctuations of NADH that result from the active site geometry (highly conserved in dehydrogenases) in a manner that enhances conformations along the transition state coordinate.
|

