 Purdue News
Purdue News
 Purdue News
Purdue News
December 1996

|
The development could speed the delivery of biotech-derived crops, experts say.
The first use of the technique in corn was announced in the December issue of Nucleic Acids Research (Vol. 24, No. 19).
Before, when scientists used a variety of methods to insert new genes into a plant's chromosomes, the insertions were random, with genes landing anywhere on the chromosomes, possibly interrupting other gene sequences that coded for vital proteins. Even if the plant survived, these random insertions have been a large concern to governmental regulators because they allowed the possibility that the genetic transformation would have unforeseen consequences.
The process will allow researchers to remove certain genes that they have inserted, such as antibiotic or herbicide-resistance genes, after the need for them has passed. "This could actually be used in all genetically engineered material released for public consumption," Hodges says.
The Purdue-developed process received a patent in June, and the technology has been licensed to Plant Genetic Systems of Gent, Belgium (a major portion of which was recently acquired by AgrEvo of Frankfurt, Germany). The company has an exclusive license for the technology in Europe and a non-exclusive license in the United States.
Peter Dunn, assistant vice president for research at Purdue and director of Purdue's Biotechnology Institute, says the technology will ease one of the central issues that has slowed the federal government's approval of the first round of genetically modified crops. "If these 'other' genes could be removed from the plant before government regulatory review -- using procedures like those recently developed at Purdue by Tom Hodges and Leszek Lyznik -- it could substantially advance the timetable for approval of transgenic crops," he says.
Ralph W.F. Hardy, president of the National Agricultural Biotechnology Council, agrees that this is a significant development in biotechnology: "The fact that this research has received a patent and is licensed indicates that this technique promises to be routine enough to have real application."
John Snyder, assistant director of Purdue's Office of Technology Transfer, says, "We've been able to demonstrate the technical feasibility to the point that companies can see the viability of the technology."
According to Hodges, the excision technique, in combination with the development of gene targeting procedures, gives researchers the opportunity to find applications in several new areas. For example, it will allow researchers to remove a defective gene from a plant, change or mutate that gene, and place the improved gene back into the plant on the same site on the chromosome as it was originally found.
|
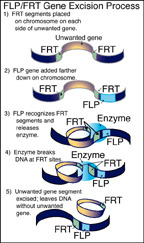
The first step in the new method is an excision technique that uses a yeast gene identified as "FLP" -- sometimes called "flip" by researchers -- and a segment of DNA that is 48 nucleotides long called "FRT."
The researchers can arrange two copies of the FRT segments on a chromosome so that they bracket the unwanted gene. Then they add the yeast gene to the mix, which produces an enzyme causing the chromosome to break at the sites of the FRT segments.
The chromosome rejoins, but without the unwanted gene, which is digested by the cell. Because the process uses the yeast FLP gene, scientists sometimes refer to such an excised gene as having been "flipped-off."
"This application is valuable from an environmental perspective," Hodges says. "Because it allows us to eliminate antibiotic-resistance genes, for example, it virtually eliminates the possibility of giving rise to the build-up of antibiotic resistance in microorganisms in the environment."
Sources: Thomas Hodges, (765) 494-4657; e-mail, hodges@btny.purdue.edu
Leszek Lyznik, (765) 494-8787; e-mail, lyznik@btny.purdue.edu
Peter Dunn, (765) 494-6840; e-mail, pedunn@ocba.purdue.edu
Ralph W.F. Hardy, home, (705) 887-9887
John Snyder, (765) 494-2610; e-mail, jrsnyder@dsp.purdue.edu
Writer: Steve Tally, (765) 494-9809; e-mail, tally@ecn.purdue.edu
Purdue News Service: (765) 494-2096; e-mail, purduenews@purdue.edu
ABSTRACT (Nucleic Acids Research, 1996, Vol. 24, No. 19)
FLP-mediated recombination of FRT sites in the maize genome
L.Alexander Lyznik, and Thomas K. Hodges, Department of Botany and Plant Pathology, Purdue University, West Lafayette, IN 47907, USA; K.V. Rao, Department of Genetics, Osmania University, Hyderbad, 50007, India
Molecular evidence is provided for genomic recombinations in maize cells induced by the yeast FLP/FRT site-specific recombination system. The FLP protein recombined FRT sites previously integrated into the maize genome leading to excision of a selectable marker, the neo gene. NPTII activity was not observed after the successful recombination process; instead, the gusA gene was activated by the removal of the blocking DNA fragment. Genomic sequencing in the region of the FRT site (following the recombination reaction) indicated that a precise rearrangement of genomic DNA sequences had taken place. The functional FLP gene could be either expressed transiently or after stable integration into the maize genome. The efficiency of genomic recombinations was high enough that a selection for recombination products, or for FLP expression, was not required. The results presented here establish the FLP/FRT site-specific recombination system as an important tool for controlled modifications of maize genomic DNA.
United States Patent Number 5,527,695
CONTROLLED MODIFICATION OF EUKARYOTIC GENOMES
Inventors: Thomas K. Hodges, Leszek Lyznik, West Lafayette, Ind.
Assignee: Purdue Research Foundation, West Lafayette, Ind.
ABSTRACT
DNA constructs are provided for the creation of transgenic eukaryotic cells. These DNA constructs allow a more precise and effective transformation procedure by enabling the targeting of DNA sequences for insertion into a particular DNA locus, while enabling the removal of any randomly inserted DNA sequences that occur as a by-product of known transformation process.
Photo captions
Purdue Professor Tom Hodges is the leader of a research team that has developed the
first commercially viable technique to eliminate unwanted genes from genetic transfers
in crops. (Purdue Agricultural Communications photo by Mike Kerper)
Color photo, electronic transmission, and Web and ftp download available. Photo ID: Hodges/Genes
Download here
The new gene excision method uses a yeast gene identified as "FLP" and a segment of
DNA that is 48 nucleotides long called "FRT." Researchers place two FRT segments
on a chromosome so that they bracket the unwanted gene. Then they add the yeast gene,
which produces an enzyme causing the chromosome to break at the FRT sites. The chromosome
rejoins, without the unwanted gene. (Agricultural Communications Service graphic
by Pam Lassiter and Steve Tally)
Color graphic, electronic transmission, and Web and ftp download available. Photo
ID:
Hodges/Genes.graphic
Download here.